
One more case for the nonpolar molecule if the molecules Nonpolar molecules are considered as a pure covalent bondīecause it forms by equal sharing of electrons between the atoms in a compoundĪnd this is what a covalent bond is known for. They are soluble in water (called hydrophilic) and other polar solvents like aceticĪcid (CH3-CO-OH), methanol (CH3-OH), etc. Polar molecules have polar covalent bonds or ionic bonds. Structure, presence of lone pair of electrons or electronegativity difference Moment and this is possible only if there is an electronegativity differenceīetween the associated atoms as well as asymmetrical geometry so that the inducedĬertain conditions for the molecule to be polar i.e. Polar molecules are those molecules that have a net dipole Polarity, let’s have some ideas about what polar and nonpolar molecule are: Here in this article, we are going to know whether XeCl2 isĪ polar or nonpolar molecule with a detailed explanation. There are many doubts regarding ‘is xenon dichloride is a trueĬompound? or it is just a van der Waals force of attraction between xenon andĬhlorine atom? because in this molecule chlorine atom is connected by a This is prepared by reacting xenon and fluorine gas together Xenon dichloride is the chemical name of XeCl2 and this is In H2O, the central atom is Oxygen atom and it is sp3 The central atom in H2S is sp3 hybridized It is connected to 5 other atoms, one more than The central atom undergoes sp3d2 hybridization. The oxygen atom in oxygen fluoride undergoes sp3Ĭarbon, the central atom, is sp2 hybridized.
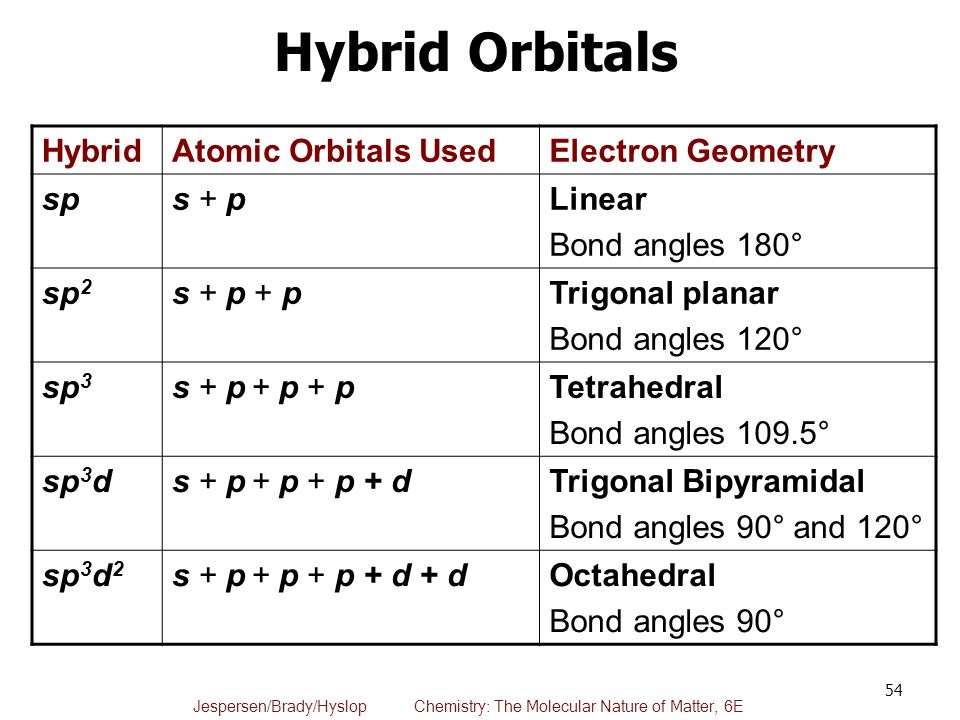
The hybridization of the carbon atom in this molecule is Hybridization of the central atom of AlH4 is sp3.

Prsumably you mean AlH4- the tetrahydroaluminate anion. The hybridization of the central atom in NOCl is sp2. Sp2,120 is the hybridization of the central atom in SO2. The central atom is oxygen with sp3 hybridization. Therefore, the number of orbitals used in the hybridization is the number of #\mathbf(sigma)# bonds made around the central atom.The hybridization in the central I atom is sp3. That is, you get five #sp^3d# orbitals, for example. You can see that the number of orbitals used in the hybridization spits out the same number of hybridized orbitals that can #sigma# bond. #sp^3d^2# hybridization in #"SF"_6# corresponds with six #sigma# bonds around one sulfur. #sp^3d# hybridization in #"PF"_5# corresponds with five #sigma# bonds around one phosphorus. #sp^3# hybridization in methane corresponds with four #sigma# bonds around one carbon. #sp^2# hybridization in ethene corresponds with three #sigma# bonds around one carbon. #sp# hybridization in acetylene corresponds with two #sigma# bonds around one carbon. One pure double bond has one #sigma# and one #pi# bond, and one pure triple bond has one #sigma# and two #pi# bonds.įrom knowing the hybridization of the central atom, we can determine the number of #sigma# bonds around the central atom, but no more than that without more information. If you want to get the number of #pi# bonds, know what compound you're looking at, and just count them.
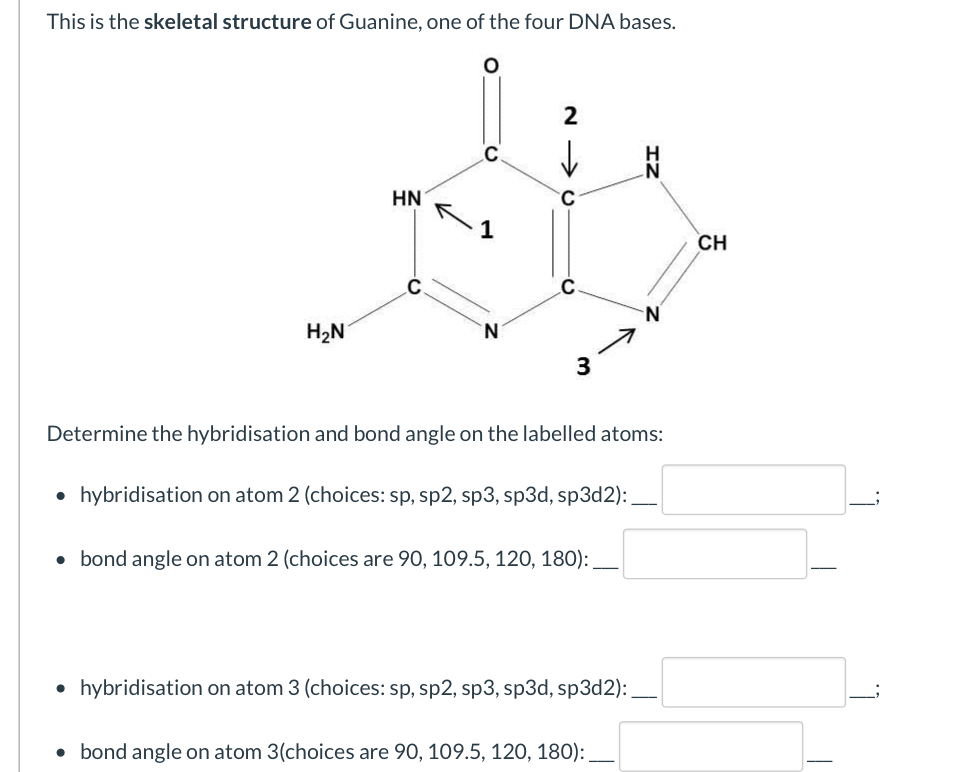
Without knowing more context, it's not possible to know the number of #pi# bonds.
